- Offerings
- Tools & Platforms
Tools & Calculators
- Open API
- Calculators
- SIP Calculator
- CAGR Calculator
- Compound Interest Calculator
- FD Calculator
- RD Calculator
- EPF Calculator
- Retirement Calculator
- HDFC SIP Calculator
- Mutual Fund Return Calculator
- Lumpsum Calculator
- Step Up SIP Calculator
- ETF SIP Calculator
- Brokerage Calculator
- Equity Margin Calculator
- SWP Calculator
- EMI Calculator
- MTF Calculator
- Markets
Stocks
F&O
Mutual Funds
- More
Symbiotec Pharmalab IPO
To be Announced
Minimum Investment
IPO Details
TBA
TBA
TBA
TBA
TBA
NSE, BSE
TBA
TBA
Symbiotec Pharmalab IPO Timeline
Bidding Start
TBA
Bidding Ends
TBA
Allotment Finalisation
TBA
Refund Initiation
TBA
Demat Transfer
TBA
Listing
TBA
Symbiotec Pharmalab Limited
Incorporated in 2002, Symbiotec Pharmalab Limited is a prominent pharmaceutical and biotechnology company specializing in the development and manufacturing of active pharmaceutical ingredients (APIs), nutritional ingredients, and specialty products. With over 30 years of industry experience, the company has evolved from a lab-scale steroidal-hormone API manufacturer into a globally recognized, backward-integrated industrial-scale platform. It holds approvals from major regulatory bodies like the US FDA and EU-GMP. Symbiotec serves domestic and international markets with a focus on research-driven manufacturing, quality compliance, and sustainable processes, catering to the pharmaceutical, nutraceutical, and wellness industries.
Symbiotec Pharmalab Limited IPO Overview
The Symbiotec Pharmalab Limited IPO is a book-built issue aiming to raise ₹2,180 crores. This comprises a fresh issue of equity shares aggregating up to ₹150 crores and an offer for sale (OFS) of up to ₹2,030 crores by existing shareholders. Key dates for the IPO bidding period, price band, and lot size are yet to be announced. The equity shares are proposed for listing on both BSE and NSE. JM Financial Ltd. is the book running lead manager, and MUFG Intime India Pvt. Ltd. is the registrar. Pre-IPO, the promoter holding stands at 34.47%. The issue structure reserves not more than 50% for QIBs, not less than 35% for retail investors, and not less than 15% for Non-Institutional Investors (NII/HNI).
Symbiotec Pharmalab Limited Upcoming IPO Details
| Category | Details |
| Issue Type | Book Built Issue IPO |
| Total Issue Size | Up to ₹2,180 Crores |
| Fresh Issue | Up to ₹150 Crores |
| Offer for Sale (OFS) | Up to ₹2,030 Crores |
| IPO Dates | TBA |
| Price Bands | TBA |
| Lot Size | TBA |
| Face Value | ₹2 per share |
| Listing Exchange | BSE, NSE |
| Shareholding pre-issue | 6,16,81,496 shares |
| Shareholding post-issue | TBA |
Symbiotec Pharmalab Limited IPO Reservation
| Investor Category | Shares Offered |
| QIB Shares Offered | Not more than 50% of the Offer |
| Retail Shares Offered | Not less than 35% of the Offer |
| NII (HNI) Shares Offered | Not less than 15% of the Offer |
Objectives of the IPO Proceeds
The Net Proceeds from the Fresh Issue are intended to be utilised as per the details provided in the table below:
| Particulars | Amount (in ₹ million) |
| Prepayment/repayment of certain outstanding borrowings | 1,125.00 |
| General corporate purposes* | [●] |
*Note: To be finalized upon determination of the Offer Price and updated in the Prospectus prior to filing with the RoC. The amount for general corporate purposes shall not exceed 25% of the Gross Proceeds.
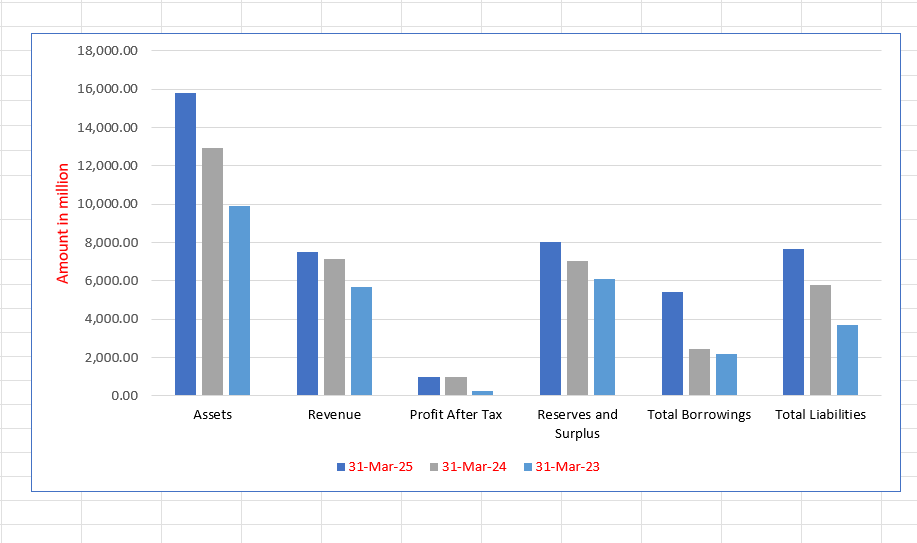
Symbiotec Pharmalab Limited Financials (in ₹ million)
| Particulars | 31 Mar 2025 | 31 Mar 2024 | 31 Mar 2023 |
| Assets | 15,796.51 | 12,947.85 | 9,899.88 |
| Revenue | 7,515.54 | 7,162.47 | 5,665.14 |
| Profit After Tax | 967.85 | 1,000.55 | 234.86 |
| Reserves and Surplus | 8,039.58 | 7,041.13 | 6,105.96 |
| Total Borrowings | 5,409.23 | 2,472.07 | 2,178.78 |
| Total Liabilities | 7,649.46 | 5,799.41 | 3,685.48 |
Financial Status of Symbiotec Pharmalab Limited
SWOT Analysis of Symbiotec Pharmalab IPO
Strength and Opportunities
- Global leadership in corticosteroid and steroidal-hormone APIs.
- Long-standing relationships with a diversified domestic and global customer base.
- Fully integrated, multi-scale manufacturing platform with backward integration.
- Clean regulatory track record with approvals from US FDA, EU-GMP, etc.
- Leading technological and R&D capabilities among Indian peers.
- Strong financial performance with high gross and EBITDA margins.
- Ability to leverage science to expand into new markets like CDMO and complex injectables.
- Entry into high-growth areas like GLP-1, Insulin, and alternative proteins via CDMO.
- First-mover advantage in developing complex products like generic Premarin and Double Chamber Vials.
- Backing by marquee investors and a seasoned leadership team with deep industry experience.
Risks and Threats
- Significant portion of gross block (53.32%) is yet to be commercialized, impacting current returns.
- High reliance on the API segment; newer verticals like complex injectables are unproven commercially.
- Increased borrowings in recent years have led to higher finance costs.
- Execution risks associated with scaling up new CDMO and complex injectables businesses.
- Dependence on key personnel and the promoter family for strategic direction.
- Intense competition from established Indian API players and Chinese manufacturers.
- Geopolitical and trade policy shifts could affect supply chains and raw material costs.
- Regulatory complexities and lengthy approval processes for new facilities and products.
- Fluctuations in prices of key raw materials like phytosterols.
- Vulnerability to foreign currency exchange rate fluctuations in export-oriented business.
Explore IPO Opportunities
Explore our comprehensive IPO pages to stay updated on the latest trends and insights.
About Symbiotec Pharmalab Limited
Symbiotec Pharmalab Limited IPO Strengths
Global Leadership in Steroidal APIs
Symbiotec Pharmalab Limited holds a commanding position in the global market for corticosteroid and steroidal-hormone APIs. With a 36.2% volume share in corticosteroids and 44.2% in steroidal-hormone APIs, it is the only company globally with a presence across the top 10 products in these categories. This leadership is built on decades of specialized expertise, backward-integrated manufacturing, and a robust regulatory track record, providing a significant competitive moat and pricing power.
Integrated and Scalable Manufacturing Platform
The company operates a fully invested, vertically integrated ‘microbe-to-pharmacy’ platform. With two operational API facilities and a newly commissioned injectables plant, it possesses in-house capabilities across fermentation (700 KL capacity) and complex chemical synthesis. This integration reduces dependency on external suppliers for key starting materials, ensures cost control, provides strategic flexibility, and forms a strong foundation for scaling its CDMO and complex injectables businesses.
Robust Research and Development Focus
Symbiotec Pharmalab Limited maintains a strong innovation ethos, continuously investing 3-4% of its revenue in R&D. It operates dedicated centres for organic chemistry, biotechnology, and complex injectables, employing over 90 scientists. This has enabled it to develop advanced capabilities like continuous flow chemistry, precision fermentation, and double-chamber vial technology, allowing it to tackle complex molecules like generic Premarin and stay ahead of the innovation curve.
Strong Financial Performance and Margins
The company has demonstrated consistent financial growth, with revenue rising at a CAGR of 15.18% from FY23 to FY25. It maintains a strong margin profile, with a gross margin of 60.43% and an EBITDA margin of 27.26% in FY25. This financial strength is supported by a favourable product mix, operational efficiencies, and supplies to regulated markets, providing the cash flow to fund future growth initiatives and ensuring financial resilience.
Experienced Leadership and Marquee Investor Backing
Symbiotec Pharmalab Limited is guided by promoters with over 30 years of industry experience and a senior management team with pedigrees from leading pharmaceutical companies. This seasoned leadership is complemented by backing from institutional investors like Rosewood Investments (InvAscent) and Motilal Oswal Alternates. This combination provides strategic vision, operational excellence, and credibility in the capital markets.
More About Symbiotec Pharmalab Limited
Company Evolution and Core Business
Symbiotec Pharmalab Limited has undergone a significant transformation since its early days in 1995 as a lab-scale manufacturer. Today, it stands as a vertically integrated pharmaceutical and biotechnology company. Its core business revolves around the development and manufacture of Active Pharmaceutical Ingredients (APIs), with a dominant focus on steroidal and hormonal compounds. The company also produces nutritional ingredients and is actively foraying into specialty products. Its operations are research-driven, emphasizing stringent quality compliance and sustainable manufacturing processes.
Operational Infrastructure and Capacity
The company’s manufacturing prowess is spread across strategic locations:
- Pithampur Facility: An established site for API manufacturing, including high-value biologics like GLP-1 analogues and Insulin.
- Ujjain Facility: A newly commissioned, multipurpose facility with 400 KL fermentation capacity. It is designed for large-volume fermentation for in-house APIs and CDMO services in food, nutraceuticals, and industrial biotechnology.
- Mhow Facility: A dedicated complex injectables plant capable of producing up to 20 million vials annually, focusing on double-chamber vials (DCVs) and other drug-device combinations.
As of June 30, 2025, the combined maximum chemical synthesis capacity was 584.67 metric tonnes, with a total fermentation capacity of 700 kilolitres.
Business Verticals and Growth Pillars
Symbiotec is strategically diversifying beyond its core API business into three interconnected verticals:
- API Products: Continues to be the cash cow, with plans to expand into classical fermentation-based APIs (e.g., immunosuppressants, anti-infectives) and complex molecules like generic Premarin.
- Contract Development and Manufacturing Organization (CDMO): Leveraging its large-scale fermentation and synthesis capabilities to offer services for pharmaceuticals (Insulin, GLP-1), nutraceuticals, alternative proteins, and industrial biotechnology. It has already secured several take-or-pay contracts.
- Complex Injectables: A forward-integration strategy into high-value drug-device combinations like DCVs, double-chamber bottles (DCBs), and double-chamber syringes (DCSs), where it claims to be a first-mover among generic companies.
Quality and Regulatory Compliance
A cornerstone of Symbiotec’s reputation is its clean regulatory track record. Its facilities hold approvals from some of the world’s most stringent regulatory bodies, including:
- United States Food and Drug Administration (US FDA)
- European Union Good Manufacturing Practices (EU-GMP)
- Ministry of Food and Drug Safety, Korea. This compliance enables it to supply to regulated markets like the US and Europe, which command better margins and foster long-term customer relationships.
Industry Outlook
Indian Pharmaceutical API and CDMO Industry
The Indian pharmaceutical industry, particularly the Active Pharmaceutical Ingredient (API) and Contract Development and Manufacturing Organization (CDMO) segments, is poised for strong growth driven by global strategic shifts and domestic capabilities.
Growth Drivers and Market Potential
- China+1 Strategy: Growing geopolitical concerns and supply chain vulnerabilities are prompting global pharma companies to diversify their API sourcing away from China. India, with its established chemical industry, skilled workforce, and cost advantages, is a primary beneficiary. This is leading to increased demand for Indian APIs and CDMO services.
- CDMO Market Expansion: The global CDMO market is projected to grow from US$ 128.8 billion in 2024 to US$ 190.2 billion by 2029, at a CAGR of 8.1%. India’s share is increasing due to significant cost advantages (30-50% lower setup costs), high-quality manufacturing standards, and a strong talent pool.
- Complex Generics and Biosimilars: The global patent cliff and the rising demand for biosimilars and complex generics (like injectables, hormonal therapies) present a large opportunity. Products with high entry barriers due to complex manufacturing or regulatory pathways offer attractive margins.
- Fermentation and Biologics Boom: There is surging global demand for fermentation-based products and biologics, including peptides like GLP-1 for diabetes/obesity and Insulin. Manufacturing these molecules requires specialized fermentation infrastructure, which is a niche capability.
Focus on Steroidal APIs and Complex Injectables
- The global market for steroidal APIs remains stable with growth driven by their use in anti-inflammatory, hormonal therapy, and critical care medicines.
- The market for complex injectables, especially differentiated drug-device combinations like Double Chamber Vials (DCVs), is high-value. DCVs command a 20-50% price premium due to advantages in stability, dosing accuracy, and reduced wastage. The addressable market for Symbiotec’s pipeline under the 505(b)(2) pathway is estimated at US$ 805.5 million in 2024, expected to grow at a 5.4% CAGR.
How Will Symbiotec Pharmalab Limited Benefit
- Benefit from Supply Chain Diversification: The global “China+1” sourcing strategy will direct increased inquiries and contracts for steroidal and fermentation-based APIs to Symbiotec, given its established scale and regulatory approvals.
- Capture CDMO Growth: Its newly commissioned, large-scale Ujjain fermentation facility positions it perfectly to capture a share of the fast-growing global CDMO market, especially in niches like alternative proteins, nutraceuticals, and biologics where it has already signed contracts.
- Monetize Complex Injectable Pipeline: As a first-mover among generics companies in backward-integrated Double Chamber Vials, it can capture significant value in this high-margin segment, with its first two products targeting a US market of US$ 204 million.
- Leverage Biologics Demand: Through its capabilities and existing agreements for Insulin and GLP-1 manufacturing, Symbiotec is well-placed to benefit from the exponential global demand for these biologic therapies.
- Expand with Proprietary Products: Successful development and commercialization of complex projects like generic Premarin (conjugated estrogens) can open up a large new revenue stream in hormone replacement therapy, leveraging its deep scientific expertise.
Peer Group Comparison
| Name of the company | Face Value (₹) | Revenue (₹ million) | Basic EPS (₹) | P/E Ratio | RoNW (%) |
| Symbiotec Pharmalab Limited | 2 | 7,515.54 | 17.70 | N.A. | 11.79% |
| Peer Group | |||||
| Concord Biotech Limited | 1 | 12,000.90 | 35.52 | 38.59 | 22.00% |
| Divi’s Laboratories Limited | 2 | 93,600.00 | 82.53 | 77.87 | 14.83% |
| Cohance Lifesciences Limited | 1 | 26,085.00 | 12.79 | 42.31 | 19.10% |
| Laurus Labs Limited | 2 | 55,540.00 | 6.65 | 152.85 | 8.00% |
Key Strategies for Symbiotec Pharmalab Limited
Expand Steroidal-Hormone API Leadership
Symbiotec Pharmalab Limited aims to solidify its global dominance in corticosteroid and steroidal-hormone APIs by broadening its portfolio. The strategy involves entering new product categories and therapeutic areas within this niche, leveraging its cost-efficient, backward-integrated model. It plans to capitalize on the supply chain shift away from China to gain market share in regulated markets like the US and EU, supported by its clean regulatory track record.
Scale Fermentation and Biologics Offerings
The company intends to build a biologics platform of scale by expanding its fermentation capabilities. This includes commercializing contracts for GLP-1 and Insulin and establishing a proposed biologics facility in Ujjain. The strategy leverages its integrated ‘microbe-to-pharmacy’ platform and multi-scale fermentation infrastructure to meet rising global demand for fermentation-based molecules, diversifying into new high-growth therapeutic categories.
Commercialize Complex Injectables Portfolio
A key forward-integration strategy involves commercializing its pipeline of complex injectables, primarily Double Chamber Vials (DCVs), through the newly commissioned Mhow Facility. Symbiotec plans to launch its first two DCV products in FY26 and is in discussions with global specialty pharma companies to license its pipeline. It is also developing related drug-device combinations (DCBs, DCSs) to enhance product differentiation in critical care settings.
Grow Diversified CDMO Services
Symbiotec Pharmalab Limited plans to scale its CDMO offerings across organic chemistry, biotechnology, and complex injectables. The strategy focuses on deepening engagements with existing clients and pursuing new opportunities in the US, EU, and other markets. It aims to leverage its Ujjain Facility for non-pharmaceutical CDMO in areas like alternative proteins and nutraceuticals, capitalizing on its scientific talent and technology platforms.
Sustain R&D and Forge Strategic Partnerships
The company will continue its R&D-focused ethos, investing in new technologies and process optimization. A core strategic pillar is to partner with large global companies for difficult-to-execute projects through risk-sharing models like milestone payments and profit-sharing arrangements. This approach aims to align long-term growth with partners, leveraging their commercial reach while Symbiotec contributes its development and manufacturing expertise.
How to apply IPO with HDFC SKY?
Follow these simple steps to apply for an IPO through HDFC SKY. Secure your investments and explore new opportunities with ease by accessing the IPOs available on the platform.
1Login to your HDFC SKY Account
2Select Issue
3Enter Number of Lots and your Price.
4Enter UPI ID
5Complete Transaction on Your UPI App
FAQs On Symbiotec Pharmalab IPO
How can I apply for Symbiotec Pharmalab IPO?
You can apply via HDFCSky using UPI-based ASBA (Application Supported by Blocked Amount).
What is the size of the Symbiotec Pharmalab IPO?
The IPO is a ₹2,180 crore offer, comprising a fresh issue of up to ₹150 crore and an Offer for Sale (OFS) of up to ₹2,030 crore.
When will the Symbiotec Pharmalab IPO open for subscription?
The IPO dates, including the opening and closing dates, are yet to be announced (TBA) by the company and lead managers.
On which stock exchanges will Symbiotec Pharmalab list?
The equity shares are proposed to be listed on both the Bombay Stock Exchange (BSE) and the National Stock Exchange (NSE).
What is the price band for the Symbiotec Pharmalab IPO?
The price band for the IPO has not been finalized yet. It will be announced closer to the IPO opening date.
What is the minimum lot size and investment for retail investors?
The lot size and consequently the minimum investment amount for retail investors are to be announced (TBA) along with the price band.
By signing up I certify terms, conditions & privacy policy

